Holobiome: Research I Articles I Updates I News I Series I Feed
Holobiome is a blog series that offers an AI-assisted summary of the latest research articles on human microbiome.
Probiotic Power-Up: Boosting Gut Harmony and Blood Sugar Balance
This new study explored how probiotics can do more than just keep the gut balanced—they may also play a role in regulating blood sugar. Researchers worked with healthy volunteers and gave some a probiotic supplement while others received a placebo over two months. They then measured the gut microbiome using shotgun metagenomics, a method that reveals which microbes are present and what functions they perform. Interestingly, the people who started with a “weaker” gut microbiome, reflected in a low wellness index, benefited the most.
One of the standout microbes was Adlercreutzia equolifaciens, which became more abundant in participants who took the probiotic. This bacterium is already known for its ability to metabolize plant compounds into helpful molecules, and in this case, its rise coincided with a stronger gut wellness score. The probiotic didn’t just shuffle microbe counts—it improved the functional side of the microbiome too. Genes linked to antibiotic resistance and virulence declined, while sugar-breaking enzymes and other beneficial pathways became more active.
The researchers also discovered that A. equolifaciens was linked to increased levels of a compound called palmitoyl serinol, which is associated with better gut wellness and healthier blood glucose levels. This suggests that the changes weren’t just superficial shifts but carried measurable metabolic benefits for the host.
To strengthen their findings, they ran a second trial using a single probiotic strain, Lacticaseibacillus casei Zhang. Remarkably, it produced similar results, again enriching A. equolifaciens and enhancing microbiome wellness. Together, these results show how probiotics may work in very targeted ways, recruiting key bacteria like A. equolifaciens that then ripple outward to support both gut and whole-body health.
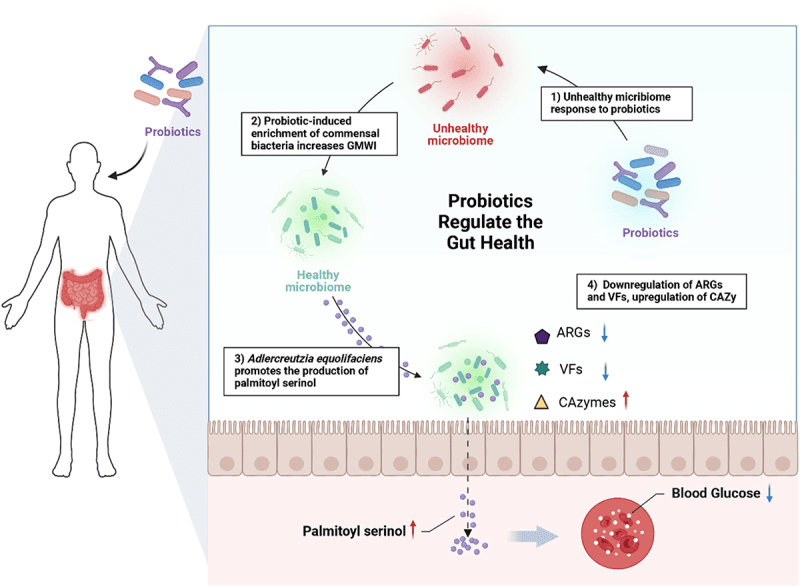
Mechanistic illustration of probiotic supplementation improving gut health in phenotypically healthy hosts.
When TB Drugs Disrupt the Gut: Understanding Microbiome Shifts During Treatment
Tuberculosis remains one of the world’s toughest infections to treat, requiring months of combination antibiotic therapy. While these drugs save lives, they also have a side effect that’s often overlooked—they disrupt the gut microbiome. In this study, researchers in eastern China followed TB patients closely throughout their treatment journey, monitoring how their gut microbes changed as the antibiotics did their work.
The results showed a clear reshaping of the gut ecosystem. Beneficial microbes, such as those linked to short-chain fatty acid production, tended to decline. These are the bacteria that normally help reduce inflammation and maintain gut lining health. At the same time, other microbial groups—some of them less desirable—found opportunities to expand. This loss of microbial balance can have consequences that extend beyond the gut, including effects on immunity and metabolism.
By looking at these shifts over time, the researchers were able to highlight that the longer and more intensive the antibiotic therapy, the greater the disruption to gut diversity. For a treatment that often lasts six months or more, this presents a challenge: while antibiotics must stay strong against TB, they may also leave the gut ecosystem vulnerable.
The findings raise an important next step for TB care—how can we protect or restore the microbiome during treatment? Whether through probiotics, prebiotic foods, or other microbiome-supportive strategies, integrating gut health into TB therapy could help patients not only beat the infection but also recover with a stronger and more balanced internal ecosystem.

Differential species analysis at the phylum level
When the Gut Talks Back to Immunotherapy
Lung cancer treatment with immune checkpoint inhibitors like pembrolizumab can be life-extending—but the gut community seems to shift along the way. In a pilot study of metastatic NSCLC patients on pembrolizumab, researchers found a clear signature of dysbiosis compared with carefully matched healthy controls. Two families that usually help make soothing short-chain fatty acids—Lachnospiraceae and Ruminococcaceae—were markedly lower in patients, while Bacteroidaceae was increased. This combination often hints at a gut, that’s less resilient and more inflamed.
Why does that matter for cancer care? Many SCFA-producing microbes (think butyrate makers) help train immune cells and keep the intestinal lining calm—a helpful backdrop when you’re trying to rally T cells against tumors. The study’s functional readouts also pointed toward fewer butyrate-linked enzyme pathways in patients, consistent with the taxonomic shifts.
The authors caution this is an opportunistic, small study without pre-treatment baselines—so the changes could reflect the cancer itself, prior chemotherapy, ongoing immunotherapy, or disease progression. Still, the signal is strong enough to fuel a practical idea: pairing immunotherapy with microbiome-supportive strategies that restore SCFA producers may be worth testing.
Zooming out, it reinforces a broader pattern seen across oncology: gut composition can track with (and sometimes shape) immunotherapy outcomes—making the microbiome less of a side note and more of a potential co-pilot in precision cancer care.
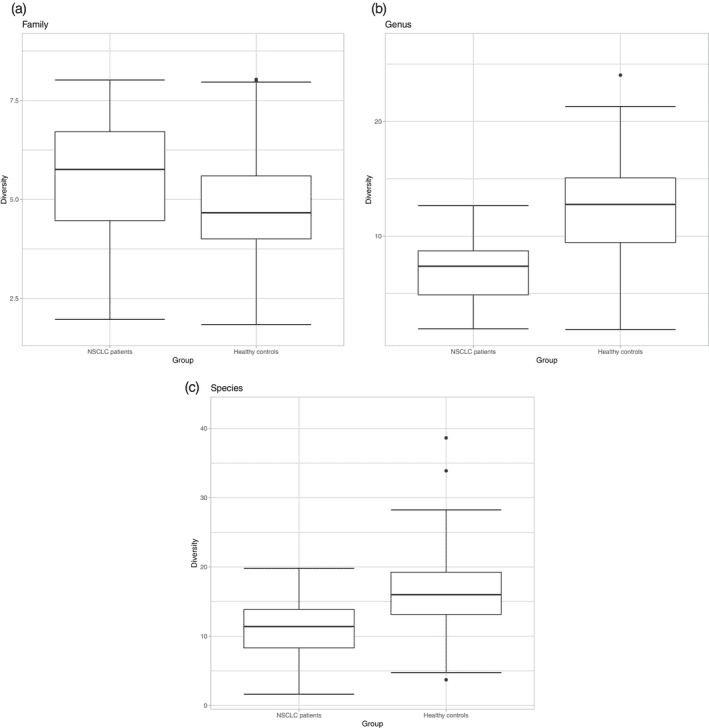
(a–c) Mean alpha diversity between NSCLC patients and healthy controls for (a) Family, (b) Genus, and (c) Species levels.
Zinc Toothpaste, Happier Plaque: Tuning the Oral Microbiome’s Metabolism
We usually think of toothpaste as something that simply cleans teeth, but this trial shows it can actively reshape the tiny community living on them. In a double-blind study with 115 adults, participants brushed for 12 weeks with either a zinc citrate trihydrate toothpaste or a regular control paste. When researchers checked the plaque microbiome, they found that brushing with zinc shifted the balance of bacteria in surprising ways.
Species of Veillonella, which feed on lactic acid and are often considered supportive commensals, increased in the zinc group. At the same time, Fusobacterium nucleatum subsp. polymorphum—a microbe linked with gum disease—decreased compared to controls. This meant zinc didn’t just sweep the surface; it shifted composition of the plaque toward a friendlier one's that may lower the risk of inflammation and periodontitis.
But the story went beyond who was present. Functional profiling of the microbial genes showed a drop in glycolysis pathways, meaning the bacteria were producing less acid—a win for enamel health. At the same time, pathways for lysine biosynthesis (an amino acid tied to gum health) and nitrate reduction (important for both oral and cardiovascular wellness) were enriched. These changes suggest that the zinc toothpaste altered not only the community members but also the chemistry of their collective metabolism.
All told, the findings highlight how oral care products can act like selective gardeners of the microbiome. Instead of simply scrubbing plaque away, zinc toothpaste seems to cultivate microbes that keep the gums calmer, teeth stronger, and possibly even contribute to systemic health through nitrate metabolism.
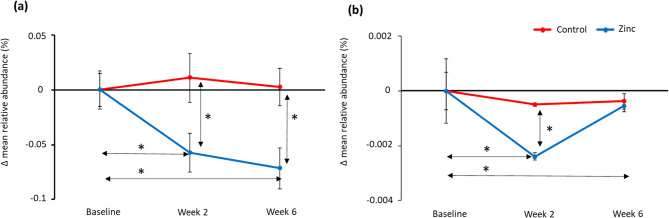
Changes in mean relative abundance (%) levels of glycolysis pathway genes for the control toothpaste (red) and zinc toothpaste (blue) compared to baseline, based on analysis using Tax4Fun (a) and meta transcriptomics (b). Significant differences (p < 0.05) are highlighted * and error bars represent standard error.
Fermented Soy Meets the Menopause Microbiome
Cheonggukjang, a traditional Korean fermented soybean dish, is gaining scientific attention for more than just its flavor. In a randomized, double-blind clinical trial of 60 postmenopausal women, researchers tested two versions of Cheonggukjang: one rich in beneficial microbes and another with fewer. Across the board, menopausal symptoms improved, but the women who consumed the high-microbe version experienced the greatest relief, especially in areas like hot flashes and overall symptom scores.
The gut microbiome appeared to play a central role in these benefits. Stool analyses showed that the high-beneficial-microbe Cheonggukjang subtly shifted the gut environment toward more balanced communities, with a trend toward a lower Firmicutes :Bacteroidetes ratio, often linked to improved gut health and metabolic resilience. Even though not all microbial shifts reached statistical significance, the direction was clear: richer ferments gave the gut a healthier profile.
The metabolic outcomes were equally telling. Only the high-microbe group showed a drop in fasting blood glucose, pointing to a potential link between the probiotics in Cheonggukjang, gut microbial functions, and host sugar metabolism. This suggests that the ferment is not just easing menopausal symptoms directly but also tweaking the gut ecosystem in ways that ripple into broader metabolic health.
What makes this study special is the emphasis on the quality of fermentation. Not all ferments are created equal, and this trial shows that ferments richer in friendly microbes may provide stronger, more consistent benefits. It’s a reminder that traditional foods like Cheonggukjang can be more than cultural staples—they may be living microbiome therapies with relevance for women’s health.
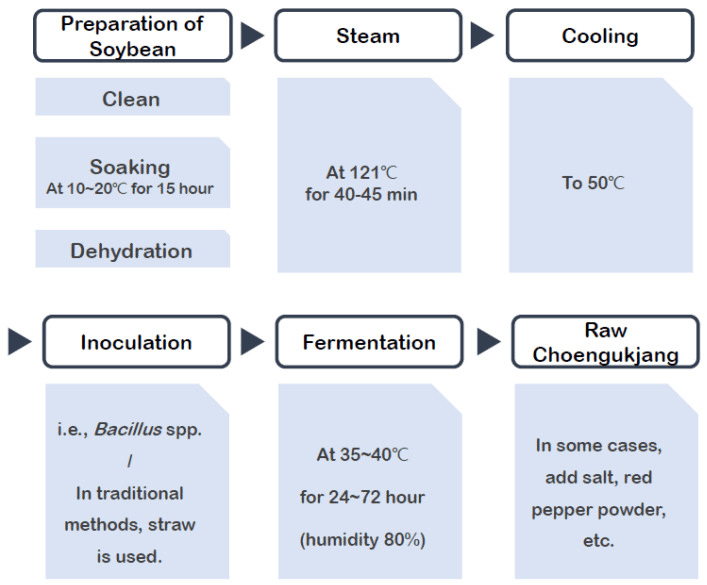
Traditional Cheonggukjang manufacturing process.
