The MicroByte – Pathogen Series: The Pathogen Behind Scarlet Fever
History
The story of Streptococcus pyogenes is closely linked to the development of germ theory and the early days of modern medicine. Diseases caused by this bacterium were recognized long before scientists understood the microbes behind them.
As early as the 4th century BCE, the Greek physician Hippocrates described a condition called erysipelas—a spreading red inflammation of the skin. While physicians could observe and document these symptoms, it took nearly two thousand years before the actual microbial cause was identified.
Discovery of Streptococcus
In 1874, Austrian surgeon Theodor Billroth observed small spherical bacteria arranged in pairs or chains in wound infections and erysipelas cases. He named these organisms “Streptococcus,” combining the Greek words streptos (chain) and kokkos (berry). This chain-like arrangement remains a defining feature of the genus today.
A few years later, in 1879, Louis Pasteur isolated these bacteria from the blood and uteruses of women suffering from puerperal fever, also known as childbed fever. His work demonstrated that streptococci were responsible for the devastating mortality rates seen in maternity wards during that time.
Naming of Streptococcus pyogenes
The formal species name Streptococcus pyogenes was introduced in 1884 by German physician Friedrich Julius Rosenbach. Studying bacteria from pus-forming infections, Rosenbach chose the name pyogenes from the Greek words pyo (pus) and genes (forming). This highlighted the bacterium’s ability to cause suppurative, or pus-producing, infections.
During the early years of bacteriology, scientists believed that different streptococcal diseases were caused by entirely different species. As a result, names such as S. erysepaltis, S. scarlatinae, and S. puerperalis were proposed based on the specific disease being observed. Later research by Frank Andrews and Alfred Christie showed that these disease-associated strains actually belonged to the same species, leading to their consolidation under the single name Streptococcus pyogenes.
The Lancefield Classification System
A major breakthrough in streptococcal classification came in the 1920s and 1930s through the work of microbiologist Rebecca Lancefield. She developed a serological classification system based on specific carbohydrate antigens present in the bacterial cell wall, known as the C-substance.
Using this system, Streptococcus pyogenes was categorized as Group A Streptococcus (GAS)—a term that is still widely used in clinical diagnostics today.
Lancefield later expanded this work in 1946 by studying the M protein, a major surface protein that plays a critical role in virulence and immune evasion. This discovery allowed scientists to subtype GAS strains.
Pathogenesis
The disease-causing ability of Streptococcus pyogenes comes from a powerful collection of virulence factors that allow the bacterium to attach to host tissues, avoid immune defenses, and damage cells. These mechanisms help the pathogen survive in very different environments in the human body, from the carbohydrate-rich surfaces of the throat to deeper tissues such as muscle.
Infection usually begins when the bacteria attach to epithelial cells of the skin or respiratory tract. This attachment is mainly driven by the M protein, the most important surface protein of S. pyogenes. It binds host molecules such as fibronectin and fibrinogen, helping the bacteria firmly anchor to host cells. Other factors strengthen this interaction. Protein F binds fibronectin and improves adhesion to epithelial cells, lipoteichoic acid helps establish the initial hydrophobic contact with host membranes, and many strains produce pili, hair-like structures that aid in attachment and biofilm formation by interacting with host receptors like gp340.
Once attached, the bacterium uses several strategies to evade the immune system. A key defense is the hyaluronic acid capsule, which surrounds the bacterial cell. Because hyaluronic acid is also found in human connective tissue, the capsule acts as molecular camouflage, helping the bacteria avoid immune detection and resist phagocytosis by neutrophils. The M protein also interferes with the complement system by binding human Factor H, preventing opsonization and reducing immune clearance. Other enzymes further weaken immune responses: C5a peptidase (ScpA) cleaves the complement component C5a, reducing neutrophil recruitment, while SpyCEP degrades interleukin-8 (IL-8), limiting neutrophil activation. In addition, S. pyogenes produces DNases that break down neutrophil extracellular traps (NETs), allowing the bacteria to escape these antimicrobial structures.
After establishing itself and avoiding immune defenses, Streptococcus pyogenes can cause tissue damage through several toxins and enzymes. Streptolysin O and Streptolysin S are cytolytic toxins responsible for the beta-hemolysis seen in bacterial culture; both form pores in host cell membranes and destroy red blood cells, white blood cells, and platelets. The enzyme streptokinase activates plasminogen to plasmin, which dissolves fibrin clots and helps the bacteria spread through tissues. Some strains also produce streptococcal pyrogenic exotoxins, such as SpeA and SpeC, which act as superantigens and trigger massive activation of T cells. This leads to a surge of inflammatory cytokines—often described as a cytokine storm—and contributes to diseases such as scarlet fever and streptococcal toxic shock syndrome.
Transmission
Streptococcus pyogenes spreads very efficiently between people, and humans are the only known natural reservoir for the bacterium. Transmission is most common during the acute stage of infection, although asymptomatic carriers—estimated to make up about 15% of the healthy population—also play an important role in maintaining its presence in communities.
The bacterium is most commonly transmitted through respiratory droplets released when an infected person coughs, sneezes, or even talks, which is why infections like strep throat spread easily in close-contact settings. It can also spread through direct contact with infected skin lesions, particularly in conditions such as impetigo or cellulitis. In some cases, the bacteria can survive briefly on contaminated objects or surfaces, including shared towels, clothing, or bedding. In addition to the throat, Group A Streptococcus can colonize other body sites such as the anus, vagina, and genital mucosa, which can serve as additional reservoirs for transmission.
The risk of spread increases in crowded environments like schools, military barracks, and long-term care facilities where close contact is common. Although uncommon, outbreaks linked to improperly handled food have occasionally been reported when food is prepared by an infected individual. Importantly, there is no evidence that pets transmit this bacterium to humans—transmission occurs strictly from person to person.
Signs and Symptoms
The diseases caused by Streptococcus pyogenes range widely in severity. They are generally grouped into superficial infections, invasive infections, and post-infectious complications that occur after the initial infection has resolved.
Superficial infections are the most common. Strep throat (streptococcal pharyngitis) typically appears suddenly with a sore throat, fever, painful swallowing, and inflamed tonsils that may have white exudates. Small red spots on the palate (palatal petechiae) may also appear, while symptoms like cough or a runny nose are usually absent. Impetigo, a common skin infection, begins as small papules that develop into pustules and eventually rupture, leaving behind thick, honey-colored crusts. Scarlet fever occurs when toxin-producing strains cause a characteristic red “sandpaper-like” rash along with a strawberry tongue, where a white coating peels away to reveal bright red papillae. Another skin infection, cellulitis, causes redness, swelling, warmth, and tenderness of the skin and typically affects deeper layers of the dermis than impetigo.
In some cases, the infection can become invasive and life-threatening. Necrotizing fasciitis, sometimes called a “flesh-eating” infection, rapidly destroys deep tissues and fascia and is often marked by severe pain that appears disproportionate to visible symptoms. Streptococcal toxic shock syndrome (STSS) is another serious condition characterized by sudden high fever, low blood pressure, and multi-organ failure, often linked to soft-tissue infections. Historically, S. pyogenes has also caused puerperal sepsis, a severe infection of the uterus that occurs shortly after childbirth and was once a major cause of maternal mortality.
Even after the initial infection has resolved, the immune system’s response to streptococcal antigens can sometimes lead to delayed complications. Acute rheumatic fever may develop after untreated throat infections and is characterized by joint inflammation, heart involvement, involuntary movements known as chorea, and skin nodules. Over time, it can cause permanent damage to heart valves. Another complication, acute post-streptococcal glomerulonephritis, affects the kidneys and may lead to swelling, high blood pressure, and dark or “cola-colored” urine due to the presence of blood.
Diagnosis
Diagnosing Streptococcus pyogenes infections usually involves a combination of clinical evaluation and laboratory testing. In cases of suspected strep throat, clinicians often look for the absence of typical viral symptoms such as cough or a runny nose, along with features more suggestive of streptococcal infection, including inflamed tonsils with exudates and tender lymph nodes. Skin infections may also be recognized based on their characteristic appearance, such as the “honey-colored” crusts of impetigo or the sharply defined redness seen in erysipelas.
Laboratory tests are used to confirm the infection. A Rapid Antigen Detection Test (RADT) can detect Group A streptococcal antigens from a throat swab within minutes, although it is slightly less sensitive than culture. The throat culture remains the gold standard, where samples are grown on blood agar and beta-hemolytic colonies are identified after 24–48 hours of incubation at about 37°C. Increasingly, nucleic acid amplification tests (NAATs or PCR) are also used because they detect bacterial DNA and offer higher sensitivity. Additional laboratory methods include bacitracin sensitivity testing, which helps distinguish S. pyogenes from other beta-hemolytic streptococci, and the PYR test, which detects the enzyme pyrrolidonyl aminopeptidase and is highly specific for this species. In severe or invasive infections, blood cultures or deep tissue samples may be required, while post-infectious complications can be confirmed using serological markers such as antistreptolysin O (ASO) and anti-DNase B antibodies that indicate a recent streptococcal infection.
Treatment
Streptococcus pyogenes remains highly sensitive to beta-lactam antibiotics, and importantly, there has been no documented clinical resistance to penicillin. For common infections such as strep throat or scarlet fever, the standard treatment is oral penicillin V or amoxicillin for about 10 days. When adherence to oral medication is a concern, a single intramuscular injection of benzathine penicillin G may be used instead.
For patients with penicillin allergies, alternative antibiotics such as cephalexin, cefadroxil, clindamycin, or macrolides like azithromycin can be prescribed. However, resistance to macrolides and clindamycin has been increasing in some regions, making careful antibiotic selection important.
Severe invasive infections require urgent medical care. Conditions such as necrotizing fasciitis or streptococcal toxic shock syndrome (STSS) are treated with high-dose intravenous penicillin G combined with clindamycin. Clindamycin is particularly valuable because it suppresses the production of bacterial toxins, which may continue to cause damage even after bacterial growth slows. In cases of necrotizing fasciitis, surgical removal of infected and dead tissue is also essential to control the infection.
Prevention
Preventing the spread of Streptococcus pyogenes largely depends on reducing transmission within communities and healthcare settings. Good hygiene practices, especially frequent handwashing with soap and water or alcohol-based sanitizers, remain the most effective preventive measure. Practicing respiratory etiquette, such as covering the mouth and nose while coughing or sneezing and disposing of tissues promptly, also helps limit the spread of respiratory droplets.
Proper wound care is equally important. Cleaning and covering skin lesions can prevent them from becoming entry points for infection or sources of bacterial spread. People diagnosed with strep throat or scarlet fever are generally advised to stay home from school or work until they no longer have a fever and have been on antibiotics for at least 12–24 hours, which significantly reduces the risk of transmission.
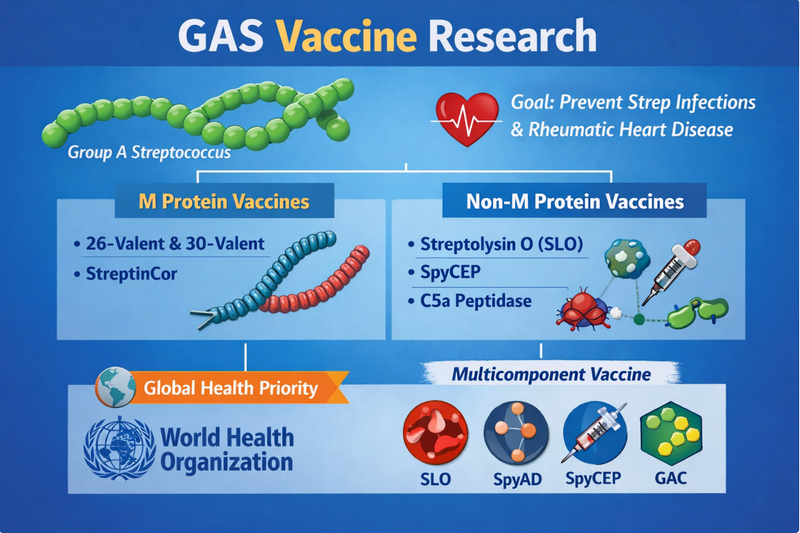
Vaccine Research
Although antibiotics are effective against Streptococcus pyogenes, researchers have long been working toward a vaccine that could prevent infection and reduce complications such as rheumatic heart disease. One major approach focuses on the M protein, the bacterium’s key surface virulence factor. Some vaccine candidates target the hypervariable N-terminal region, which requires including multiple strain types to achieve coverage. Examples include the 26-valent and 30-valent vaccines, designed to protect against the most common circulating serotypes. Other candidates, such as StreptInCor, instead target the conserved C-terminal region of the M protein in an effort to provide broader protection across many strains.
Researchers are also exploring non-M protein vaccines that target other conserved bacterial components. These include surface proteins and toxins such as Streptolysin O (SLO), SpyCEP, and C5a peptidase, which play important roles in immune evasion and tissue damage. One notable example is the four-component vaccine candidate developed by GlaxoSmithKline, which combines SLO, SpyAD, SpyCEP, and Group A carbohydrate (GAC) to stimulate a broader immune response. Because of the global burden of diseases like rheumatic heart disease, the World Health Organization has identified the development of a Group A Streptococcus vaccine as a major global health priority.
Microbe Profile
Shape: Spherical to ovoid cocci arranged in pairs or chains
Gram nature: Gram-positive
Spore formation: No
Biofilm formation: Yes
Oxygen requirement: Facultative anaerobe (Aerotolerant)
Optimal temperature: 37℃ (Growth possible from 15℃ to 40℃)
Optimal pH: Approximately 7.4 to 7.5
Nutrient Usage: Multiple amino acid auxotroph; requires blood/serum enriched media
Taxonomic classification
Domain: Bacteria
Phylum: Bacillota (formerly Firmicutes)
Class: Bacilli
Order:Lactobacillales
Family:Streptococcaceae
Genus:Streptococcus
Species:Streptococcus pyogenes

-Neha Rao
Also Read: The MicroByte- Pathogen Series: Entamoeba histolytica-An ancient parasite
References
Etymologia: Streptococcus. (2016). Emerging Infectious Diseases, 22(11), 1977. https://doi.org/10.3201/eid2211.ET2211
Walker, M. J., Barnett, T. C., McArthur, J. D., Cole, J. N., Gillen, C. M., Henningham, A., Sriprakash, K. S., Sanderson-Smith, M. L., & Nizet, V. (2014). Disease manifestations and pathogenic mechanisms of Group A Streptococcus. Clinical microbiology reviews, 27(2), 264–301. https://doi.org/10.1128/CMR.00101-13
Brouwer, S., Rivera-Hernandez, T., Curren, B. F., Harbison-Price, N., De Oliveira, D. M. P., Jespersen, M. G., Davies, M. R., & Walker, M. J. (2023). Pathogenesis, epidemiology and control of Group A Streptococcus infection. Nature reviews. Microbiology, 21(7), 431–447. https://doi.org/10.1038/s41579-023-00865-7
Wang, J., Ma, C., Li, M., Gao, X., Wu, H., Dong, W., & Wei, L. (2023). Streptococcus pyogenes: Pathogenesis and the Current Status of Vaccines. Vaccines, 11(9), 1510. https://doi.org/10.3390/vaccines11091510
Gera, K., & McIver, K. S. (2013). Laboratory growth and maintenance of Streptococcus pyogenes (the Group A Streptococcus, GAS). Current protocols in microbiology, 30, 9D.2.1–9D.2.13. https://doi.org/10.1002/9780471729259.mc09d02s30
Loughman, J. A., & Caparon, M. (2006). Regulation of SpeB in Streptococcus pyogenes by pH and NaCl: a model for in vivo gene expression. Journal of bacteriology, 188(2), 399–408. https://doi.org/10.1128/JB.188.2.399-408.2006
Savic, D. J., & McShan, W. M. (2012). Long-term survival of Streptococcus pyogenes in rich media is pH-dependent. Microbiology (Reading, England), 158(Pt 6), 1428–1436. https://doi.org/10.1099/mic.0.054478-0