The MicroByte Pathogen Series: Listeria monocytogenes- The Intracellular Invader
History
A hundred years ago, in 1926, EGD Murray and his colleagues published a characterisation of a bacterium they had isolated from laboratory rabbits that had died from previously undescribed causes– calling this microbe Bacterium monocytogenes , for the higher than usual monocytes they found in the blood of the affected animals . The genus Listeria was described later in 1940 by Pirie, named to commemorate the renowned surgeon and pioneer of antiseptic practices, Joseph Lister . Another notable name in the history of this microbe is HPR Seeliger, who wrote ‘Listeriosis’, and collected thousands of microbial isolates from a variety of environments in order to further study it .
For a long time in its scientific history, L. monocytogenes was considered zoonotic (i.e, mainly transmitted from infected animals) until food-associated outbreaks were reported in the 1980s by Schlech and group . Later food related outbreaks further confirmed that Listeria monocytogenes is a food-borne pathogen, the way we know it today . We now know that L. monocytogenes is a major cause for concern in the production and storage of food, since it can survive refrigeration temperatures, and can survive in several niches like cheeses, dairy, meats, and even food processing facilities, soil, and even excreta. Epidemics of listeriosis have been documented, and thousands of cases continue to occur every year.
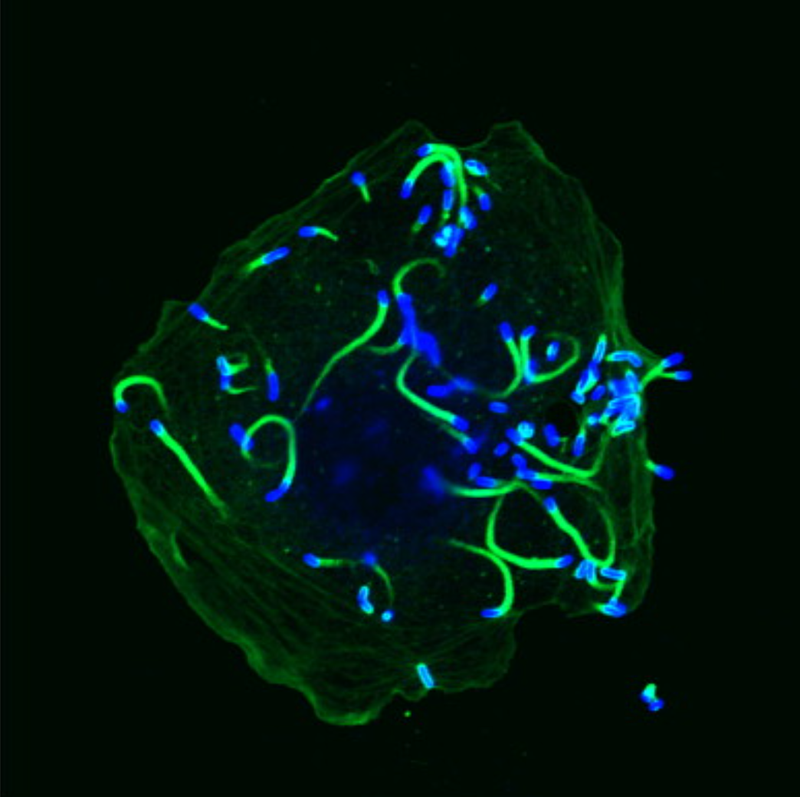
Electron micrograph showing an infected cell with Listeria bacteria labelled blue, with their actin assemblies labelled in green
Transmission and Pathogenesis
A key feature of L. monocytogenes that has led to it being a model organism for cellular biology is its ability to invade and reside in host cells, and even spread from cells to their neighbours without being detected by the host immune system. Its journey into host cells requires an arsenal of biological tools, or ‘virulence factors’ that are encoded into its genome and whose functions are used in a concerted mechanism to complete a successful invasion and further propagation. L. monocytogenes from the external extracellular environment possesses surface molecules aptly named internalins which interact with receptors (signal receiver molecules) on the host cell, triggering it to take in the microbe. Other molecules have also been found to help mediate this process, helping in a variety of the subprocesses involved in bacterial internalisation such as adhesion and even the exposure of the internalins on the bacterial surface.
When it enters the host cell, it is immediately present inside what is known as a phagocytic vacuole (compartment formed by the modification of the host cell membrane, as a result of phagocytosis, meaning the engulfing of an external body by a cell). Here, its next set of virulence factors come into play– listeriolysin O and phospholipases. L. monocytogene’s trademark toxin, listeriolysin O perforates the membrane of the phagocytic vacuole, allowing phospholipases, capable of further breaking down the vacuole’s membrane components to pass through, together facilitating the complete entry of L. monocytogenes into the host cell. Listeriolysin O has also been shown to help in host invasion in multiple other related ways such as disruption of host protein synthesis, and suppressing the cell’s oxidative stress response.
Here, a protein actA allows it to take cellular structural components in the form of filaments known as actin, and polymerize them at one end of the microbe, which drives this whole assembly around the host cell, eventually reaching its boundaries, where it protrudes into the neighbouring host cell. The neighbouring cell then takes in these protrusions, the microbe once again dissolves the original host cell’s membranes and then the neighbouring cell’s membranes, allowing it to continue the infection cycle unexposed to the host immune system. In intestinal goblet cells, after the first host cell is invaded, this microbe translocate directly into lamina propria, a deeper layer of connective tissue from where it can propagate through the system.
On a macro level, Listeria monocytogenes can be found in a myriad of sources in the environment, but human illness and outbreaks are mostly associated with contaminated food. It is also present as part of the gastrointestinal microbiome in controlled levels.
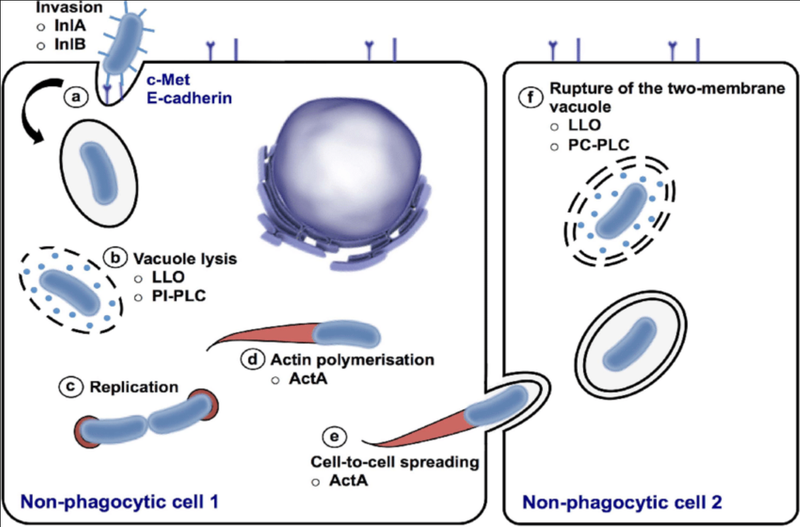
The mechanism of the intracellular invasion of host cells by L. monocytogenes
Signs and Symptoms
In healthy, immunocompetent individuals, infection with L. monocytogenes causes gastrointestinal illness which runs its course, and typically self resolves. However, it is more dangerous in vulnerable populations like immunocompromised patients, the elderly, newborns, or those who are pregnant. Complications of L. monocytogenes infection include encephalitis, meningitis, spontaneous abortion, and sepsis.
Diagnosis
To diagnose an L. monocytogenes infection, a CSF, blood, or placental fluid sample is required, which gives a positive culture result for the same.
Treatment
The treatment of Listeria monocytogenes infections is accomplished through antimicrobial agents, such as penicillin G or ampicillin administered intravenously. However, due to observed instances of antibiotic resistance in this microbe, treatments must be customised according to patient specific resistance patterns.
Prevention
At a population level, preventing the spread of Listeria monocytogenes requires sanitary food processing and preparation, and subsequent safe storage. Appropriate personal hygiene measures like hand-washing are also necessary to be maintained. Proper vigilance and specific and speedy detection of L. monocytogenes in prepared food materials, as well as effective cleaning methodology for the respective facilities could be useful tools in fighting future spread of this infection.
Microbe Profile
Gram status: +ve
Shape: Bacillus/ small rods
Spore formation: No
Motile: Both intracellular and extracellular motility
Oxygen requirements: Facultative anaerobe
Optimum temperature: 30–37°C (growth)
Optimum pH: 6-7
Taxonomic Classification
Domain: Bacteria
Kingdom: Bacillati
Phylum: Bacillota
Class:Bacilli
Order: Bacillales
Family:Listeriaceae
Genus:Listeria
Species:Listeria monocytogenes
-Antara Arvind
Also Read: The MicroByte Pathogen Series: Bordetella pertussis- Bacterium Behind Whooping Cough
References
Abdulkadieva, M. M., Sysolyatina, E. V., Vasilieva, E. V., Litvinenko, V. V., Kalinin, E. V., Zhukhovitsky, V. G., Shevlyagina, N. V., Andreevskaya, S. G., Stanishevskyi, Y. M., Vasiliev, M. M., Petrov, O. F., & Ermolaeva, S. A. (2023). Motility provides specific adhesion patterns and improves Listeria monocytogenes invasion into human HEp-2 cells. PLoS ONE, 18(8), e0290842. https://doi.org/10.1371/journal.pone.0290842
Cheng, C., Yang, Y., Dong, Z., Wang, X., Fang, C., Yang, M., Sun, J., Xiao, L., Fang, W., & Song, H. (2015). Listeria monocytogenes varies among strains to maintain intracellular pH homeostasis under stresses by different acids as analyzed by a high-throughput microplate-based fluorometry. Frontiers in Microbiology, 6, 15. https://doi.org/10.3389/fmicb.2015.00015
Cossart, P. (2007). Listeriology (1926–2007): the rise of a model pathogen. Microbes and Infection, 9(10), 1143–1146. https://doi.org/10.1016/j.micinf.2007.05.001
Etymologia:Listeria. (2016). Emerging Infectious Diseases, 22(4), 633. https://doi.org/10.3201/eid2204.et2204
Hof, H. (2003). History and epidemiology of listeriosis. FEMS Immunology & Medical Microbiology, 35(3), 199–202. https://doi.org/10.1016/s0928-8244(02)00471-6
Matereke, L. T., & Okoh, A. I. (2020). Listeria monocytogenes Virulence, Antimicrobial Resistance and Environmental Persistence: A Review. Pathogens, 9(7), 528. https://doi.org/10.3390/pathogens9070528
Osek, J., & Wieczorek, K. (2022). Listeria monocytogenes—How this pathogen uses its virulence mechanisms to infect the hosts. Pathogens, 11(12), 1491. https://doi.org/10.3390/pathogens11121491
Pizarro-Cerdá, J., & Cossart, P. (2018). Listeria monocytogenes: cell biology of invasion and intracellular growth. Microbiology Spectrum, 6(6). https://doi.org/10.1128/microbiolspec.gpp3-0013-2018
Roberts, B. N., Chakravarty, D., Gardner, J., Ricke, S. C., & Donaldson, J. R. (2020). Listeria monocytogenes Response to Anaerobic Environments. Pathogens, 9(3), 210. https://doi.org/10.3390/pathogens9030210
Rogalla, D., & Bomar, P. A. (2023, July 4). Listeria monocytogenes. StatPearls - NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK534838/