The MicroByte- Pathogen Series: Entamoeba histolytica-An ancient parasite
History
Entamoeba histolytica has evolved since prehistoric times. Paleontological evidence suggests that E. histolytica may have originated in Western and Southern Europe during the New Stone Age. Historical records from Chinese, Hebrew, and Greek sources describe the symptoms of dysentery; however, there is no specific mention of E. histolytica as the causative agent.
ELISA analyses of fossilized feces and samples collected from ancient drainage and sewage systems have confirmed the presence of E. histolytica. Traces of the parasite were identified in Israel and dated back to medieval times. This finding suggests that E. histolytica may have originated in Europe and was later introduced to Israel by European travelers. However, E. histolytica has also been identified in archaeological sites in North America dating back to the pre-Columbian era, indicating that the parasite was already present in North and South America in prehistoric times. It is believed to have spread through soil.
In the 19th century, Entamoeba histolytica was formally recognized. Physicians of the modern era documented the symptoms of amoebic dysentery and other associated manifestations of infection. However, they were initially unable to identify the causative agent or determine the underlying cause of the disease.
In 1875, the Russian medical doctor Fedor Aleksandrovich Lösch was the first to provide a detailed description of amoebic dysentery. He not only documented the symptoms but also regularly analyzed the stool samples of a patient. He observed that the patient’s stool was thin, reddish-brown in color, foul-smelling, and contained mucus and pus-classic signs of dysentery. He also noted the intermittent shedding of amoebae in the stool. Furthermore, he observed that when amoebae were absent from the stool, there was an improvement in enteritis and stool consistency. The patient also exhibited symptoms of pleuritis. Unfortunately, the patient died due to exhaustion and anemia. During the post-mortem examination, Lösch observed severe inflammation and thickening of the mucosal membrane of the large intestine. He described the morphology of the amoeba in detail through drawings in his publications and named it Amoeba coli. However, he believed that Amoeba coli was not responsible for the inflammation and instead thought that it inhabited the intestine during the later stages of the disease.
In 1883, the German physician and microbiologist Robert Koch visited India and Egypt during a cholera epidemic. During this period, he investigated five cases of dysentery, two of which involved liver abscesses. Based on his observations, he inferred that the amoeba might be responsible for the disease outcomes, contrary to Lösch’s earlier conclusion.
Naming
In 1891, the American pathologist William Thomas Councilman and the Canadian physician and parasitologist Henri Amadée Lafleur were the first to report that amoebic dysentery presented with a distinct set of clinical symptoms and was caused by a specific pathogen. They named the pathogen Amoeba dysenteriae.
While the discovery of the causative agent of amoebic dysentery was occurring in different regions of the world, the organism was referred to by different names. For example, Europeans called the disease-causing organism Amoeba coli, whereas in America it was termed Amoeba dysenteriae. This discrepancy created confusion regarding its proper classification and nomenclature.
Finally, the German zoologist Fritz Schaudinn proposed the name Entamoeba histolytica for the disease-causing species. The term “histolytica” means “tissue-dissolving,” referring to its ability to lyse and damage host tissues. Entamoeba dispar is morphologically identical to E. histolytica; however, the key difference between the two species is that E. dispar is non-pathogenic and does not cause disease, whereas E. histolytica is capable of causing invasive disease.
Pathogenesis
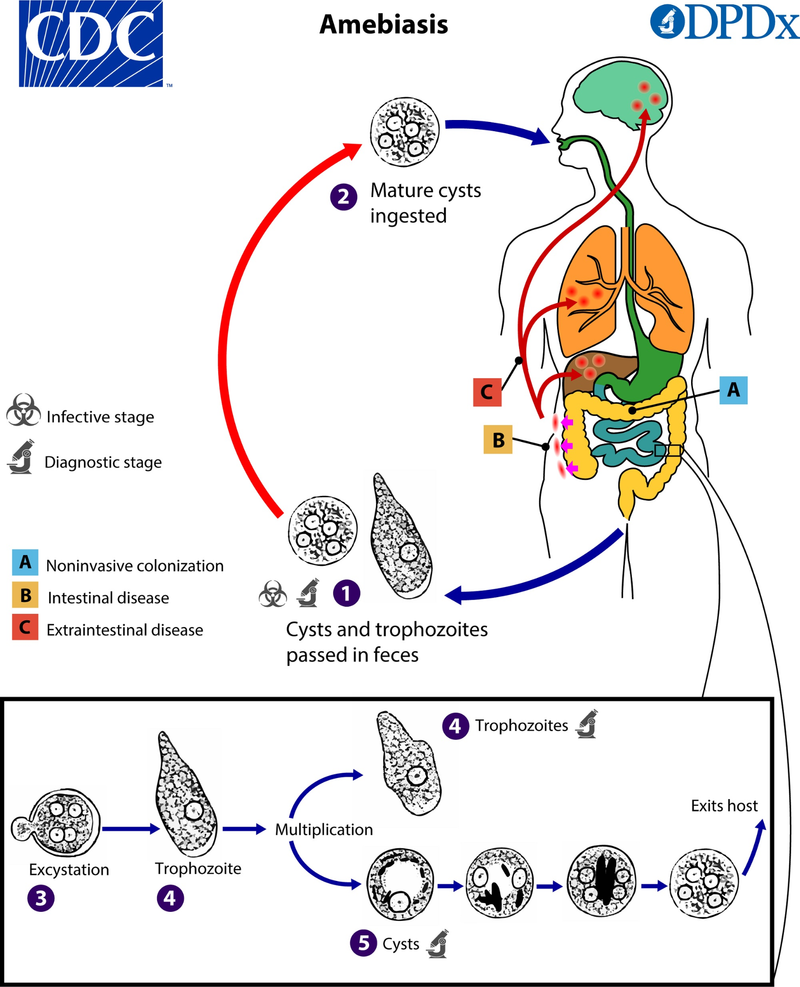
The life cycle of Entamoeba histolytica is biphasic, meaning that the parasite exists in two forms: the trophozoite and the cyst. The trophozoite form is responsible for the invasion of intestinal epithelial cells and may disseminate to other organs, leading to clinical symptoms of infection. In contrast, the cyst form is dormant and excreted in the stool. This form is responsible for transmission to a new host. However, the mechanisms that regulate the transformation between the trophozoite and cyst stages remain unclear.
Infection with Entamoeba histolytica is often asymptomatic, with only about 10% of infected individuals developing clinical symptoms. It has been speculated that certain genetic variations, such as single nucleotide substitutions in specific regions of the parasite’s DNA, may contribute to its virulence. However, further studies are required to identify definitive genetic markers associated with virulence among different isolates of E. histolytica.
Studies indicate that the pathogenic form of E. histolytica employs several strategies to establish infection.
Invasion of the Parasite
The trophozoites produce several enzymes that facilitate cellular invasion, including:
Cysteine protease A5 (CP-A5):
A multifunctional enzyme that degrades colonic mucus, remodels structural proteins such as collagen to facilitate tissue invasion, and contributes to tissue damage leading to liver abscess formation. It also interacts with receptors on goblet cells (mucus-producing cells) in the intestine and induces pro-inflammatory responses.
Cysteine proteinase CP112:
This enzyme degrades proteins responsible for maintaining cell-to-cell adhesion, resulting in tissue damage and increased intestinal permeability. These effects contribute to the symptoms of amoebic dysentery.
Cell Killing and Immune Evasion
The parasite acquires nutrients by killing host cells. E. histolytica attaches to host cells and ingests fragments of them, leading to cell death. To evade detection by the host’s immune system, the parasite displays host-derived proteins from the ingested cell fragments on its surface, thereby avoiding immune recognition and preventing the activation of immune responses.
Transmission
Transmission of Entamoeba histolytica is via the fecal-oral route. Ingestion of food or water contaminated with the cysts of Entamoeba histolytica is the one of the major causes of amoebic dysentery. It is suggested that the faeces of animals like dogs, pigs and monkeys can act as a reservoir, nonetheless there has been no substantial evidence for this.
Signs and Symptoms
Most of the time, infection by Entamoeba histolytica is asymptomatic. However, there is a chance of 10% of asymptomatic individuals to develop symptoms.
Entamoeba histolytica mostly causes intestinal amoebiasis; it can rarely cause extraintestinal infections of the liver, brain, heart and respiratory tract.
Symptoms of Gastrointestinal infections: Diarrhea, bloody stools, weight loss, and abdominal pain.
Symptoms of Liver infection: Fever, right upper quadrant pain, hepatomegaly and hepatic tenderness. Few of the patients also suffer from jaundice.
Symptoms of Respiratory infection: Fevers, coughing, and respiratory distress
Symptoms of Cardiac infection: Pericarditis or cardiac tamponade.
Symptoms of Brain infections: Headache and vomiting.
Diagnosis
The conventional method for diagnosing amoebic dysentery was stool microscopy, which was used to identify the trophozoite stage of the parasite. However, this method cannot distinguish between pathogenic and non-pathogenic species.
In recent years, molecular diagnostic techniques such as polymerase chain reaction (PCR), immunoassays including enzyme-linked immunosorbent assay (ELISA), and serological tests for the detection of specific antigens have been employed to confirm the presence of pathogenic species. These methods are often used in addition to microscopic examination for diagnostic accuracy.
Treatment
The first-line of treatment for intestinal amebiasis and amebic liver abscess is metronidazole. Metronidazole kills the active state of the parasite–the trophozoites. It is then followed by luminal agents like diloxanide furoate, paromomycin and diiodohydroxyquin. The luminal agents help to eliminate cysts and trophozoites, which aids in prevention of relapse of infection. Alternative medications to metronidazole include nitazoaxanide, tinidazole and ornidazole.
Prevention
Maintaining proper hand hygiene, avoiding raw fruits, vegetables and undercooked meats or fish can reduce the risk of parasitic infections. It is best to avoid drinking water from taps or wash fruits and vegetables from tap water especially in patients who are immunocompromised.
Microbe profile
Shape: Trophozoite: irregular, ameboid and amorphous.
Cyst: Spherical
Oxygen requirement: Microaerophilic parasite
Optimal temperature: 35.5 to 37 C
Optimal pH: 6.5- 7
Laboratory Media: Diamond’s Media
Taxonomic classification
Domain: Eukaryota
Class: Archamoebae
Order:Mastigamoebida
Family:Entamoebidae
Genus:Entamoeba
Species: Entamoeba histolytica
-Khushi.C
Also Read: The MicroByte Pathogen Series: Listeria monocytogenes- The Intracellular Invader
References
Steverding D. (2025). The history of entamoebiasis. Parasitology, 152(6), 563–572. https://doi.org/10.1017/S0031182025100279
Chou, A., & Austin, R. L. (2023, April 17). Entamoeba histolytica Infection. StatPearls - NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK557718/
Olivos-García, A., Saavedra, E., Nequiz, M., Santos, F., Luis-García, E. R., Gudiño, M., & Pérez-Tamayo, R. (2016). The oxygen reduction pathway and heat shock stress response are both required for Entamoeba histolytica pathogenicity. Current genetics, 62(2), 295–300. https://doi.org/10.1007/s00294-015-0543-5
Gillin, F. D., & Diamond, L. S. (1980). Entamoeba histolytica and Entamoeba invadens: Effects of temperature and oxygen tension on growth and survival. Experimental Parasitology, 49(3), 328–338. https://doi.org/10.1016/0014-4894(80)90069-7
Ortíz-Estrada, G., Calderón-Salinas, V., Shibayama-Salas, M., León-Sicairos, N., & de la Garza, M. (2015). Binding and Endocytosis of Bovine Hololactoferrin by the Parasite Entamoeba histolytica. BioMed research international, 2015, 375836. https://doi.org/10.1155/2015/375836
Dans, L. F., & Martínez, E. G. (2007). Amoebic dysentery. BMJ clinical evidence, 2007, 0918.
Guillén N. (2023). Pathogenicity and virulence of Entamoeba histolytica, the agent of amoebiasis. Virulence, 14(1), 2158656. https://doi.org/10.1080/21505594.2022.2158656