Holobiome: Research I Articles I Updates I News I Series I Feed
Holobiome is a blog series that offers an AI-assisted summary of the latest research articles on human microbiome.

Fine-Tuning the Gut Ecosystem in Parkinson’s Disease
In recent years, scientists have increasingly turned their attention to the gut in trying to understand neurological disorders, and this clinical trial adds another piece to that puzzle in Parkinson’s disease. People with Parkinson’s frequently experience gastrointestinal symptoms—especially constipation—years before motor symptoms appear. Mounting evidence suggests that gut dysbiosis, including reduced levels of short-chain fatty acid (SCFA)–producing bacteria and shifts in inflammatory microbial taxa, may influence both intestinal barrier integrity and systemic inflammation. This study explored whether a multi-strain probiotic could gently reshape that altered microbial ecosystem and improve clinical outcomes.
Participants with Parkinson’s disease were given either a probiotic formulation containing Lacticaseibacillus rhamnosus, Lactobacillus acidophilus, Lactiplantibacillus plantarum, and Enterococcus faecium or a placebo for twelve weeks. Stool sequencing revealed that probiotic supplementation did not dramatically overhaul overall microbial diversity—suggesting the gut ecosystem remained broadly stable—but it did selectively enrich certain beneficial taxa. Notably, there was an increase in Blautia faecicola and members of the Odoribacteraceae family, groups associated with SCFA production and bile acid metabolism. These metabolic functions are important because SCFAs such as butyrate help maintain gut barrier integrity and modulate immune responses, while bile acid transformations can influence both microbial ecology and host signaling pathways.
Alongside these microbial shifts, participants receiving the probiotic showed reductions in inflammatory markers such as TNF-α and improvements in constipation and some motor and non-motor symptoms. While the study does not prove causation, it suggests that targeted microbial modulation may dampen inflammatory signaling and enhance gut function, potentially influencing the gut–brain axis in Parkinson’s disease.
Overall, the findings highlight a key principle in microbiome science: even modest, selective changes in specific bacterial groups—rather than sweeping alterations in diversity—may be enough to shift metabolic outputs and inflammatory tone. In conditions like Parkinson’s, where gut and brain are deeply intertwined, carefully designed probiotic strategies could become part of a broader effort to support neurological health through microbial ecology.

Differential abundance analysis showing differences in community composition between microbial populations in active and placebo groups (top family level, middle genus level, bottom species level—top 25 most abundant taxa). The obtained scatter plots classify bacterial taxa into four different categories based on their abundance in compared groups: (A) not significant and not biologically relevant (gray), (B) biologically relevant, but not statistically significant (green), (C) statistically significant, but not biologically relevant (blue), and (D) biologically and statistically significant (red). Positive fold changes indicate enrichment in active group (right‐hand side), negative fold changes indicate enrichment in the placebo group (left‐hand side). FC, fold changes. [Color figure can be viewed at wileyonlinelibrary.com]
Microbial Fermentation and Mineral Absorption: A Prebiotic Study
In this randomized, double-blind crossover trial, scientists took a close look at how prebiotic fibers — specifically inulin-type fructans — interact with the gut microbiome in people living with type 2 diabetes (T2D), and whether those interactions might ripple out to influence bone health. This isn’t your typical bone-density study; the focus here is on the microbial ecology of the gut and its metabolic output. Prebiotics like inulin escape digestion in the upper gut and become food for fiber-fermenting microbes in the colon. When gut bacteria break down these fibers, they produce short-chain fatty acids (SCFAs) such as butyrate and propionate, compounds that have far-reaching effects on intestinal pH, mineral solubility, immune signaling, and even systemic inflammation — all of which are mechanisms that could link the microbiome to nutrient absorption and bone remodeling.
Participants alternated between six-week periods of inulin supplementation and a control (maltodextrin), with washouts in between, while researchers measured serum calcium, magnesium, vitamin D, and bone turnover markers. Although prebiotic supplementation did not produce statistically significant improvements in these bone-related biomarkers compared with the control, the study’s microbial lens revealed something intriguing: changes in gut microbial metabolites and community dynamics appeared to correlate with shifts in host markers, hinting at a gut–host cross-talk that may influence bone physiology.
What makes this trial particularly engaging for the science-curious is that it acknowledges the microbiome not as a passive backdrop but as an active metabolic organ. The fermentation of inulin by gut microbes — rather than the fiber itself — is what generates SCFAs and other metabolic signals that can modulate local gut conditions and possibly impact mineral absorption and immune modulation. These microbial processes can lower intestinal pH, enhance the solubility of minerals like calcium and magnesium, and influence immune mediators such as TNF-α and IL-6, all of which have been linked in other research to bone turnover and strength.
Although the primary clinical outcomes were not significantly improved in this short intervention, the study underscores a key microbiome insight: dietary fibers act through the microbes that consume them, and understanding that metabolic interplay — and how it varies person-to-person — will be essential in designing future interventions that truly harness the microbiome to support systemic health, including potentially bone metabolism in chronic conditions like T2D.
Overview of the study design. Abbreviation: ITF, inulin-type fructans
Rewriting Health Through the Lens of the Microbiome
In this clinical trial, researchers took an unconventional microbiome angle on a very human problem: postoperative sleep disturbance (PSD). Rather than focusing solely on the nervous system or pain pathways, the team examined whether the complex oral microbiota—the diverse community of microbes colonizing saliva and mouth surfaces—might be linked with sleep quality in surgical patients, and whether an anesthetic-related drug could shift that microbial landscape to improve outcomes. PSD affects many patients after surgery and can worsen recovery, pain sensitivity, and inflammation. Intraoperative infusion of esketamine, a faster-acting, more potent isomer of ketamine, was tested against placebo in people with preexisting sleep disorders to see if it could reduce PSD and, intriguingly, whether those effects might align with differences in oral microbes.
The trial confirmed that patients receiving esketamine had a lower incidence of sleep disturbance and better subjective sleep quality soon after surgery compared with controls, as measured by standard scales (NRS and AIS). While the overall richness and diversity of the oral microbiota didn’t differ dramatically between groups, specific taxa did. On the first postoperative day, individuals in the esketamine group had higher relative abundances of Streptococcus and Peptostreptococcus at the genus level and Prevotella nigrescens at the species level, alongside a lower proportion of Bacteroidota overall. These subtle shifts are notable because Prevotella and Streptococcus are among common commensals in the oral cavity that can engage with host immune signaling and mucosal health, while changes in Bacteroidota often reflect broader ecosystem restructuring.
Correlational analyses further suggested connections between certain oral bacteria—like Neisseria, Capnocytophaga, and Haemophilus species—and poorer sleep scores in some patient subgroups. That doesn’t prove causation, but it does support a growing idea in microbiome research: microbial communities in the mouth and gut can produce metabolites and immune signals that traverse mucosal barriers, interact with neural and endocrine systems, and potentially influence systemic states like sleep quality. Esketamine itself has known anti-inflammatory and neuro-modulatory effects, and this trial opens the door to a fascinating hypothesis—that part of its benefit might involve modulating microbiota-linked inflammatory or signaling pathways, offering an early glimpse of the oral microbiome’s role in sleep physiology.
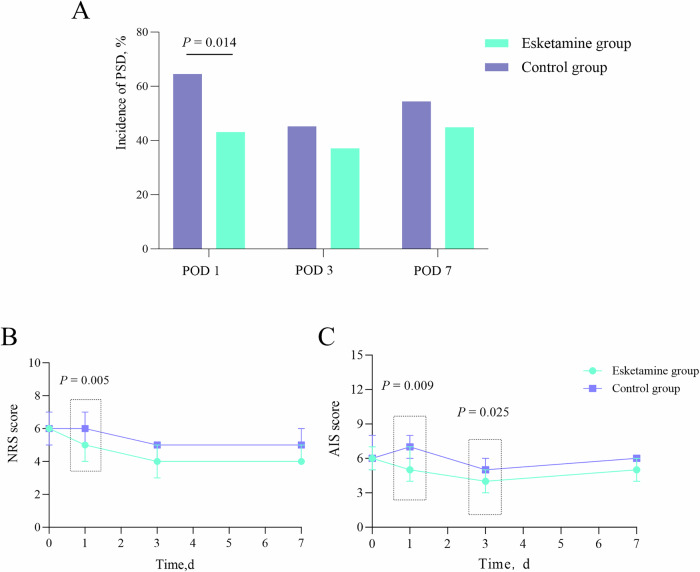
PSD Postoperative Sleep Disturbance, POD Postoperative Day, NRS Numerical Rating Scale, AIS Athens Insomnia Scale.
Nutritional Beginnings and the Microbial Blueprint of Health
The first years of life are a critical period for the establishment and maturation of the gut microbiome, a dynamic ecosystem of bacteria that helps educate our immune system, extract nutrients from food, and influence metabolism throughout life. This new study asked a provocative question: could nutritional exposures early in life leave a lasting imprint on the gut microbiota that persists into later childhood and possibly shape health outcomes years down the road? To explore this, the researchers followed a cohort of children who had been part of a nutritional intervention early in life and compared their gut microbial profiles at 3 years and again at 9 years old. What they found suggests that early dietary influences might indeed shape the trajectory of microbial community development, with possible long-term ramifications.
When scientists looked at the composition of the gut microbiota across these time points, they observed persistent differences in specific taxa among those who received early nutritional intervention versus those who did not. Some beneficial bacterial species that were more abundant in the intervention group at 3 years—such as fiber-degrading and short-chain fatty acid–producing microbes known to support gut and immune health—remained distinctive even by age 9, despite the overall community shifting as children grew. This suggests that early microbial “seeding” and dietary shaping can leave an ecological footprint that endures beyond infancy, potentially influencing metabolism, immunity, and disease susceptibility later in childhood.
The shifting microbial patterns over time also illustrate an important theme in microbiome research: although the diversity and dominant taxa in the gut evolve with age, diet, and environment, early life exposures can establish baseline differences that persist. These differences might steer the microbiome toward health-promoting configurations—rich in bacteria that ferment complex carbohydrates into beneficial metabolites like short-chain fatty acids—or toward less favorable states, depending on nutritional context and other influences.
For science-curious readers, this study highlights a powerful microbiome insight: the gut microbiome is highly malleable in early life but becomes increasingly stable with age, meaning that nutritional and microbial exposures during infancy and early childhood may act like developmental cues with long-lasting effects on microbial ecology and host physiology.
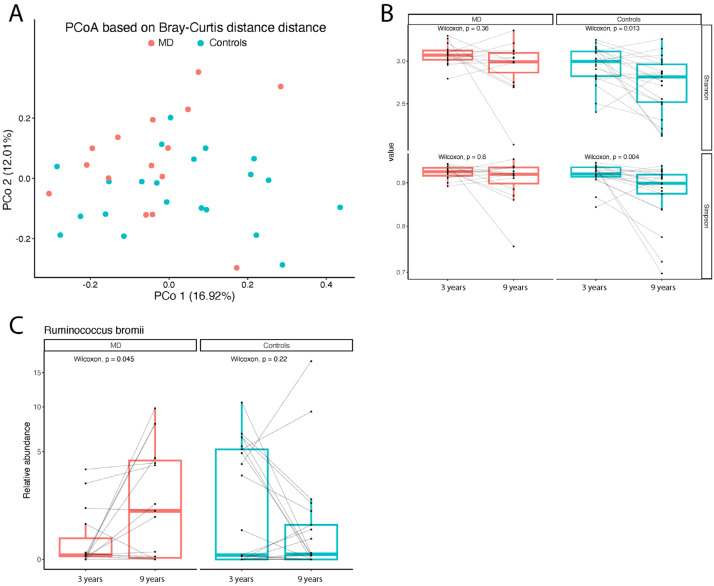
Analysis of gut microbiota composition. (A) PCoA plane based on Bray–Curtis distance showing differences in the intestinal microbiota between MD and controls at nine years. Each point represents a sample. (B) Boxplots showing the longitudinal within-group alpha diversity of the gut microbiota. Each point represents a sample, and the horizontal gray lines connect samples from the same subject. (C) Boxplot comparing the relative abundance (%, on the y-axis) of Ruminococcus bromii between samples collected from the same subjects at different timepoints (3 and 9 years). Each point represents a sample. The horizontal gray lines connect samples from the same subject.
Microbial Flexibility: How the Gut Adapts to Different Meats
In this randomized cross-over study, researchers explored a question that often sparks debate in nutrition science: does the type of meat we eat meaningfully reshape our gut microbiota? Healthy young adults alternated between diets centered on lean red beef (Pirenaica breed) and lean white meat (chicken), allowing each participant to serve as their own control. Rather than focusing solely on cholesterol or weight, the study zoomed in on the gut ecosystem—tracking how different protein sources influence microbial composition and function.
Overall, the findings suggest that both lean red and lean white meat can fit into a balanced diet without dramatically disrupting the gut microbiome in healthy individuals. Microbial diversity—a key marker of gut resilience—remained largely stable across both dietary phases. However, subtle compositional shifts did emerge. Periods of lean beef consumption were associated with relative changes in certain protein- and bile-tolerant bacterial groups, including microbes commonly linked to amino acid fermentation and bile acid metabolism. In contrast, the chicken-based phase appeared to maintain or slightly favor microbes associated with carbohydrate fermentation and short-chain fatty acid production, though differences were modest.
Importantly, these microbial shifts were not indicative of dysbiosis or inflammatory imbalance. Instead, they reflected the microbiome’s metabolic flexibility—its ability to adapt to different substrates reaching the colon. Protein type influences the profile of peptides, amino acids, and fat that escape digestion and become fuel for gut bacteria. These substrates, in turn, shape microbial metabolic outputs, including branched-chain fatty acids and other fermentation byproducts.
For science-curious readers, the takeaway is nuanced: in healthy young adults, swapping lean beef for chicken does not radically transform the gut microbiome. Rather than a dramatic overhaul, the microbiota exhibits fine-tuned adjustments based on dietary inputs—highlighting its remarkable adaptability and reinforcing that overall dietary pattern may matter more than a single protein source.
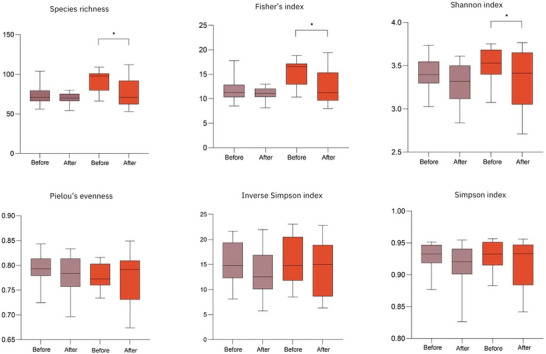
Alpha diversity indices of the intervention and control groups. The data are expressed as the median and interquartile range according to the Wilcoxon test. Richness and Fisher's index, which quantify the relationship between the number and abundance of species, were calculated. For microbial diversity and evenness, Shannon, Pielou, Simpson, and inverse Simpson's indices were performed. Pirenaica breed beef‐based diet group (n = 16); chicken‐based diet group (n = 16). Box plots represent the interquartile range (25th–75th percentile); the line inside within each box indicates the median, and the whiskers extend to minimum and maximum values. * Indicates statistically significant differences (p < 0.05).